43 label each carbon atom with the appropriate geometry
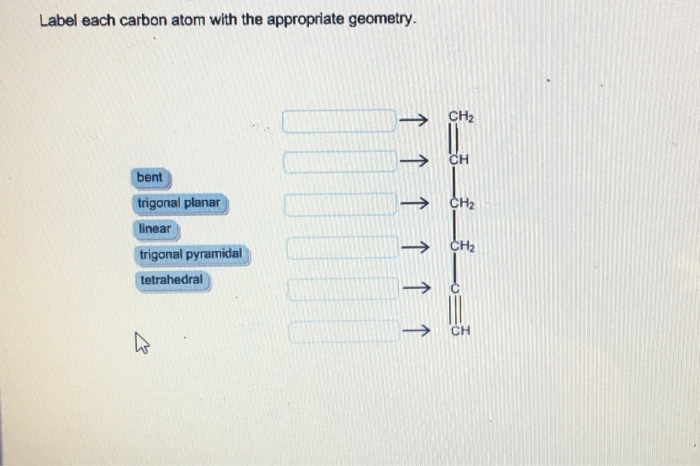
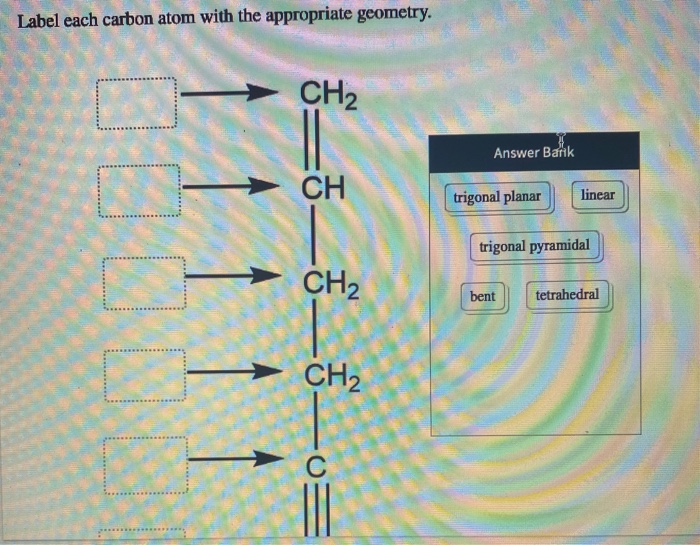
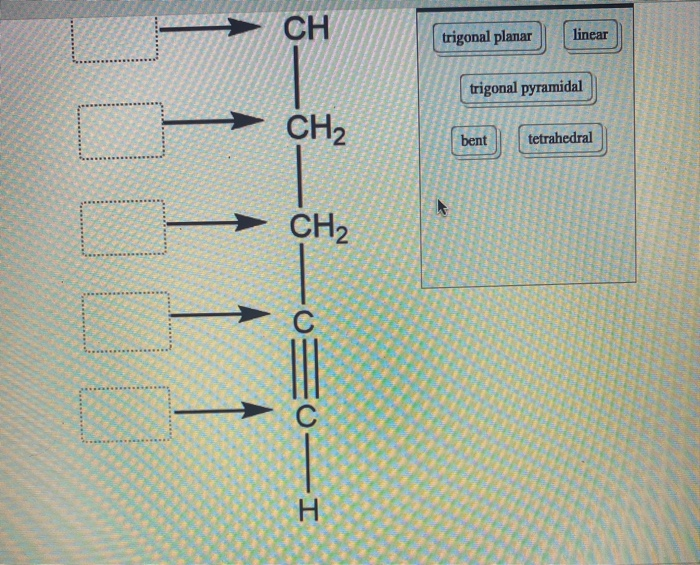
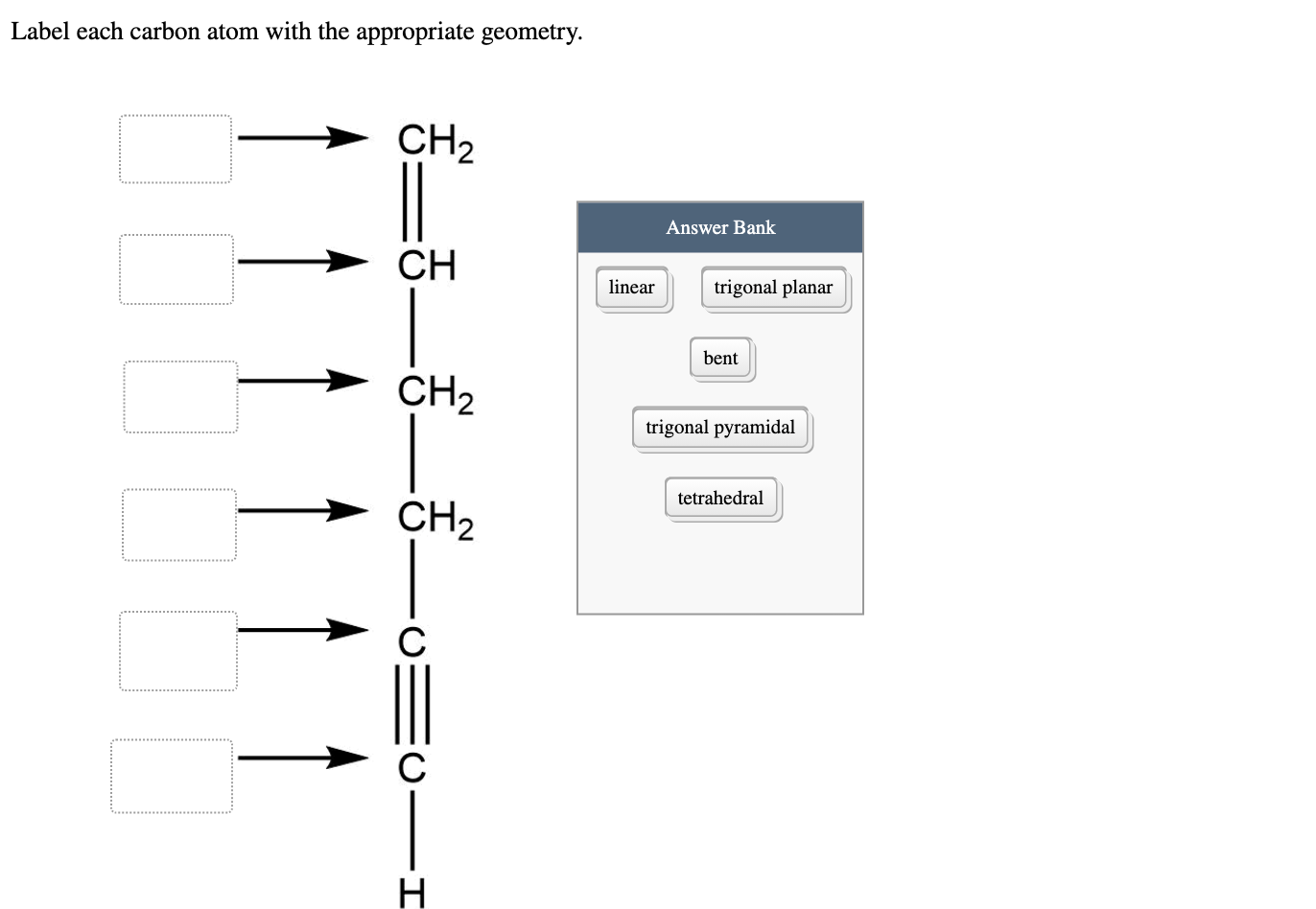
Label each carbon atom with the appropriate geometry. Label each carbon ... Label each carbon atom with the appropriate geometry. Answer Concepts and Reason Before they overlap with the 1st orbital of hydrogen, the first atomic orbitals undergo hybridization to create hybrid orbitals. Hybrid orbitals of carbon are formed through hydrogen bond formation. The type of hybridization determines the geometry at each carbon. Answered: Label each carbon atom with the… | bartleby Transcribed Image Text: Label each carbon atom with the appropriate geometry. CH2 Answer Bank CH tetrahedral linear trigonal planar bent CH2 trigonal pyramidal CH2 C C H C -I Expert Solution Want to see the full answer? Check out a sample Q&A here See Solution star_border Students who've seen this question also like: Chemistry
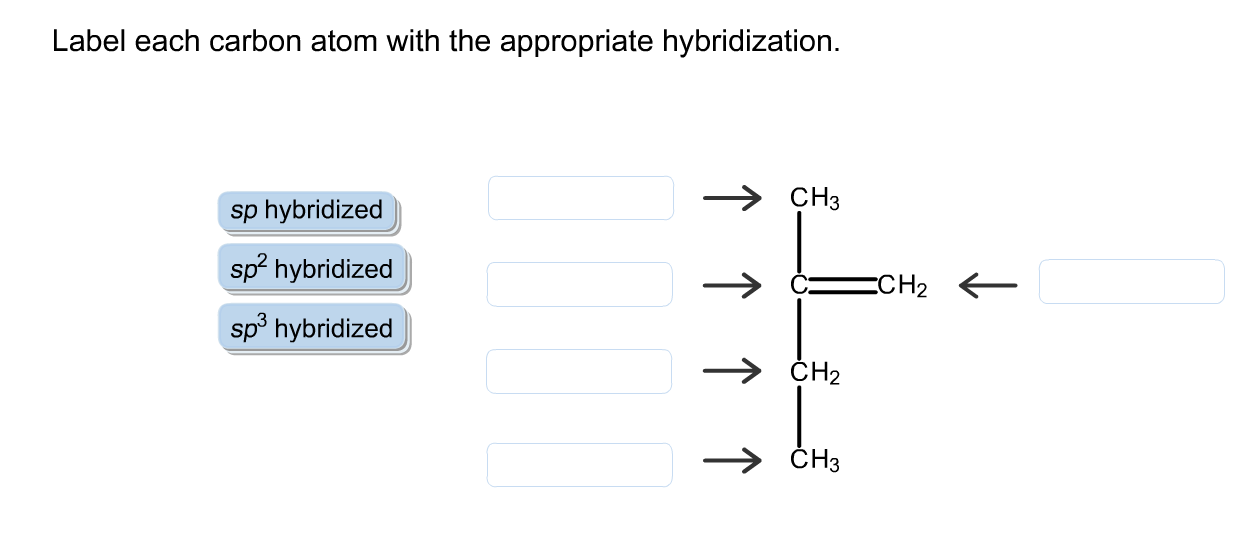
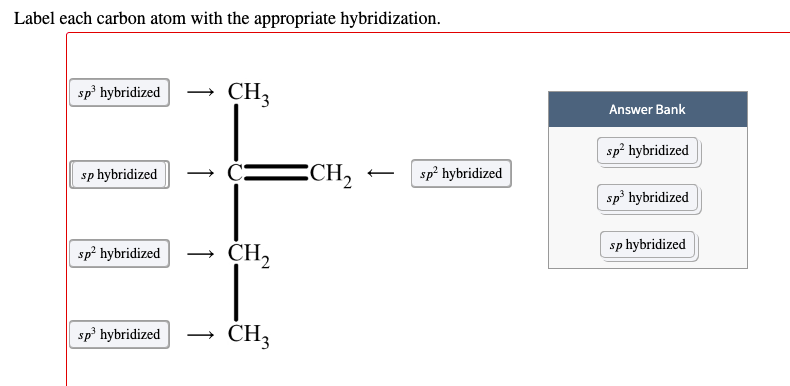
Solved Label each carbon atom with the appropriate | Chegg.com Label each carbon atom with the appropriate hybridization. sp,sp2 or sp3 Question : Label each carbon atom with the appropriate hybridization. sp,sp2 or sp3 This problem has been solved!

Label each carbon atom with the appropriate geometry
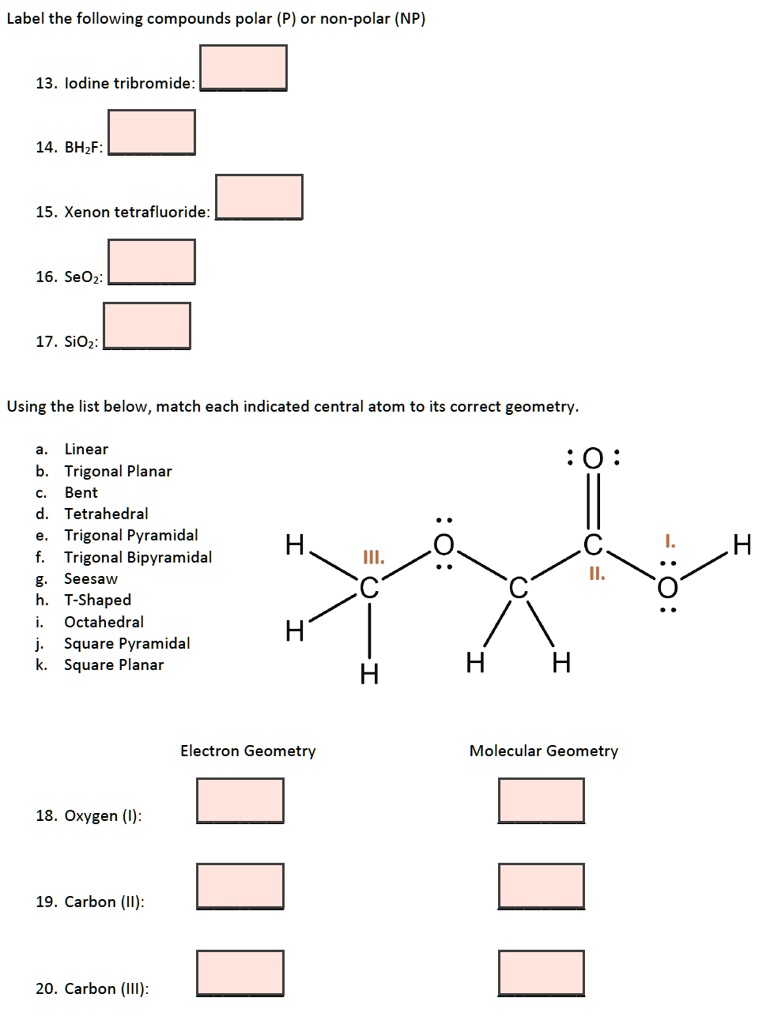
Answered: What two atomic orbitals or hybrid… | bartleby Q: Predict the approximate molecular geometry around each carbon atom of acetonitrile. H3C-C=N: The… A: When carbon is attached by 4 single bond with other atom , then the hybridization of carbon is SP3 .… Question : Label each carbon atom with the appropriate geometry. - Chegg Expert Answer. 97% (78 ratings) Transcribed image text: Label each carbon atom with the appropriate geometry. What is the geometry around each of the three central atoms in the CH ... Carbon 2. This atom has three atoms directly attached and no lone pairs. Its electron geometry and its molecular geometry are both trigonal planar. Oxygen 3. This atom has two atoms directly attached and two lone pairs. Its electron geometry is tetrahedral but its molecular geometry is bent as in water. (From Meritnation)
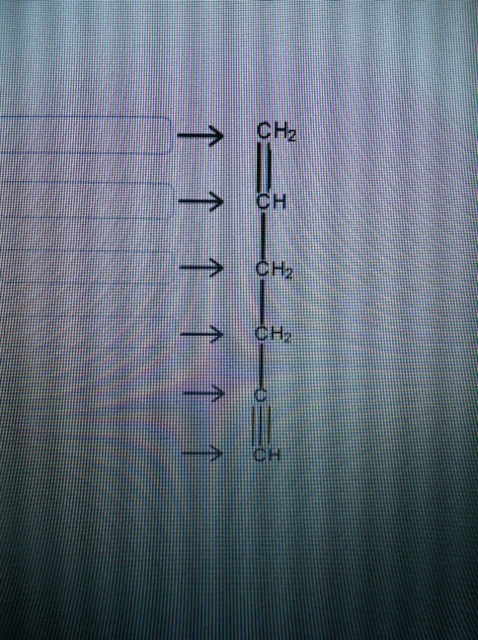
Label each carbon atom with the appropriate geometry. Label each carbon atom with the appropriate geometry. - OneClass 5 Nov 2019 Label each carbon atom with the appropriate geometry. Trigonal pyrimidal Trigonal planar Tetrahedral Linear Bent CH2 (double bond) CH (single bond) CH2 (single bond) CH2 (singlebond) C (triple bond) CH Show full question + 20 Watch For unlimited access to Homework Help, a Homework+ subscription is required. Bunny Greenfelder Lv2 Finding the hybridization of atoms in organic molecules (worked ... AP Chem: SAP‑4 (EU), SAP‑4.C (LO), SAP‑4.C.3 (EK), SAP‑4.C.4 (EK) We can find the hybridization of an atom in a molecule by either looking at the types of bonds surrounding the atom or by calculating its steric number. In this video, we use both of these methods to determine the hybridizations of atoms in various organic molecules. Question : Label each carbon atom with the appropriate geometry. - Chegg The top carbon has three things bonded to it, so its general formula is AB3, or TRIGON… View the full answer Transcribed image text : Label each carbon atom with the appropriate geometry. Solved Label each carbon atom with the appropriate geometry. | Chegg.com This problem has been solved! Who are the experts? Experts are tested by Chegg as specialists in their subject area. We review their content and use your feedback to keep the quality high. Transcribed image text: Label each carbon atom with the appropriate geometry. CH2 Answer Bank CH linear trigonal pyramidal CH2 tetrahedral trigonal planar ...
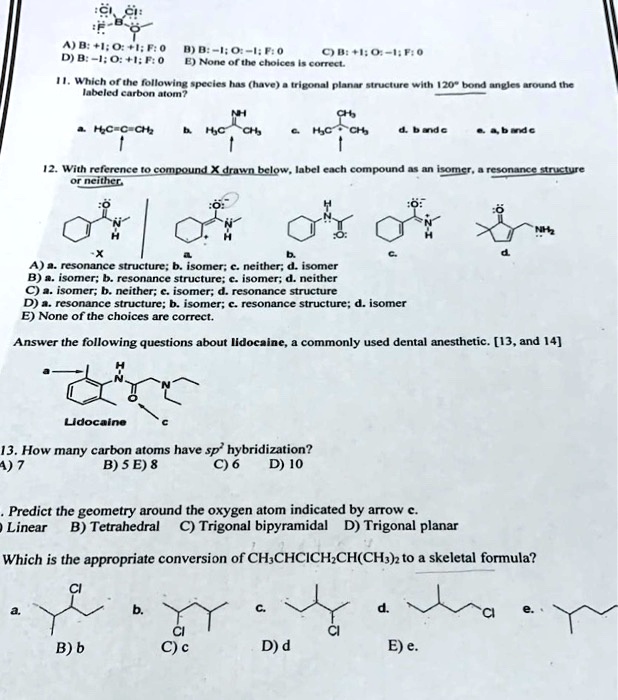
OChem Spring 2017 Exam 1 Flashcards | Quizlet 6 carbons? hexane Draw kekule structure of Methane Draw the kekule structure of Butane Draw the line structure of Butane Carbon must have _____ bonds 4 Draw the kekule structure of CH₃CHClCH₃ Draw the line structure of CH₃CHClCH₃ (2 configurations) Draw the kekule structure for CH₃CH (CH₃)CH₂CH₃ Name structure (cover right side of screen) Methane Label each carbon atom with the appropriate geometry. - Transtutors Label each carbon atom with the appropriate geometry. Trigonal pyrimidal Trigonal planar Tetrahedral Linear Bent CH2 (double bond) CH (single bond) CH2 (single bond) CH2 (single bond) C (triple bond) CH Q: Q: Label each carbon atom with the appropriate geometry. Answered: 5. Label each asymmetric carbon in the… | bartleby Question. If you are accepting the question then solve the all parts otherwise I will downvote..... Transcribed Image Text: 5. Label each asymmetric carbon in the compound below as R or S OH H CH, CH, Br Br 6. Would a 50:50 mixture of (2R,3R)-2,3-dibromobutane and (2R,35)-2,3-dibromobutane be optically active? Solved Label each carbon atom with the appropriate geometry. | Chegg.com Question: Label each carbon atom with the appropriate geometry. trigonal pyramidal CH₂ trigonal pyramidal CH tetrahedral CH₂ tetrahedral CH₂ linear C linear C Incorrect エー Answer Bank tetrahedral trigonal planar linear bent trigonal pyramidal This problem has been solved! See the answer why is this wrong? Show transcribed image text Expert Answer
Name the geometry around each carbon atom. What is the hybridization of ... Below is the Lewis structure of cyclohexane (C 6 H 12) molecule, a cyclic compound used in the manufacture of nylon and found in the distillation of petroleum.. Name the geometry around each carbon atom. What is the hybridization of each carbon atom? Chapter 9 Homework Flashcards - Questions and Answers | Quizlet There are 6 C atoms in the molecule. Starting on the left, the hybridizations are: sp2, sp2, sp3, sp, sp, sp3. All single bonds are bonds. Double and triple bonds each contain 1 bond. This molecule has 8 C-H bonds and 5 C-C bonds, for a total of 13 bonds. Double bonds have 1 bond and triple bonds have 2 bonds. This molecule has a total of 3 bonds. Label each carbon atom with the appropriate geometry. - Transtutors Label each carbon atom with the appropriate geometry. Bent trigonal pyramidal trigonal planar tetrahedral linear Recent Questions in Chemistry Q: Create a titration curve (on graph paper) for the reaction of 25.0 mL of 0.050 M Ti3+ and 0.0195 M C6H402 (quinone). The solution is 1.0 M in HCIO4 throughout the titration. label each carbon atom with the appropriate hybridization 🚀To book a personalized 1-on-1 tutoring session:👉Janine The Tutorhttps://janinethetutor.com🚀More proven OneClass Services you might be interested in:👉One...
Answered: Predict the approximate molecular… | bartleby Predict the approximate molecular geometry around each carbon atom of acetonitrile: H3C C N: right C atom left C atom linear linear bent bent trigonal planar trigonal planar trigonal pyramidal trigonal pyramidal tetrahedral tetrahedral. ... Label each carbon atom with the appropriate geometry. CH2 Answer Bank CH tetrahedral linear trigonal…
Label each carbon atom with the appropriate geometry. - SolutionInn Label each carbon atom with the appropriate geometry. trigonal planar CH2 trigonal planar CH trigonal planar linear tetrahedral ČH2 tetrahedral bent tetrahedral ČH2 trigonal pyramidal linear bent CH ↑ ↑ ↑ ↑ Expert Answer In general Hybridisation Geometry Sp3 Tetr… View the full answer This problem has been solved! See the answer
Carbon Bonds Overview & List | How Many Bonds Does Carbon Make ... Label each carbon atom with its optimum C - C - C bond angle. 120 degrees 109.5 degrees 90 degrees 180 degrees (Image) Predict the geometry around the carbon centers in the following molecule.
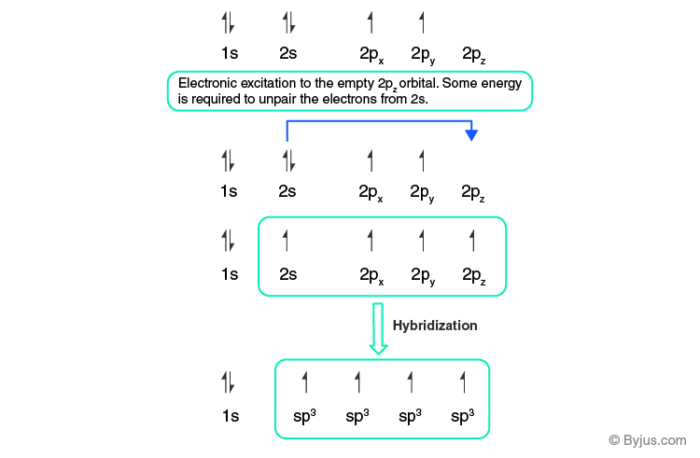
[Solved] Label each carbon atom with the appropriategeometry ... The hybrid orbitals of carbon involve in bond formation with hydrogen. Hence, the geometry at each carbon depends on the type of hybridization. Fundamentals The geometry of sp3 hybridized carbon atom is tetrahedral. The geometry of sp2 the hybridized carbon atom is trigonal planar. The geometry of sp hybridized carbon atom is linear.
⚗️Label each carbon atom with the appropriate geometry. Bin 1 points to ... Explanation: A single C-C or C-H bond is in a tetrahedral geometry, the carbon atom is bonded to four species with a bond angle of 109°. A C=C bond is trigonal planar with a bond angle of 120°. Lastly, a C≡C bond has a linear geometry with a bond angle of 180° between the atoms of the bond.
What is the geometry around each of the three central atoms in the CH ... Carbon 2. This atom has three atoms directly attached and no lone pairs. Its electron geometry and its molecular geometry are both trigonal planar. Oxygen 3. This atom has two atoms directly attached and two lone pairs. Its electron geometry is tetrahedral but its molecular geometry is bent as in water. (From Meritnation)
Question : Label each carbon atom with the appropriate geometry. - Chegg Expert Answer. 97% (78 ratings) Transcribed image text: Label each carbon atom with the appropriate geometry.
Answered: What two atomic orbitals or hybrid… | bartleby Q: Predict the approximate molecular geometry around each carbon atom of acetonitrile. H3C-C=N: The… A: When carbon is attached by 4 single bond with other atom , then the hybridization of carbon is SP3 .…
























Post a Comment for "43 label each carbon atom with the appropriate geometry"